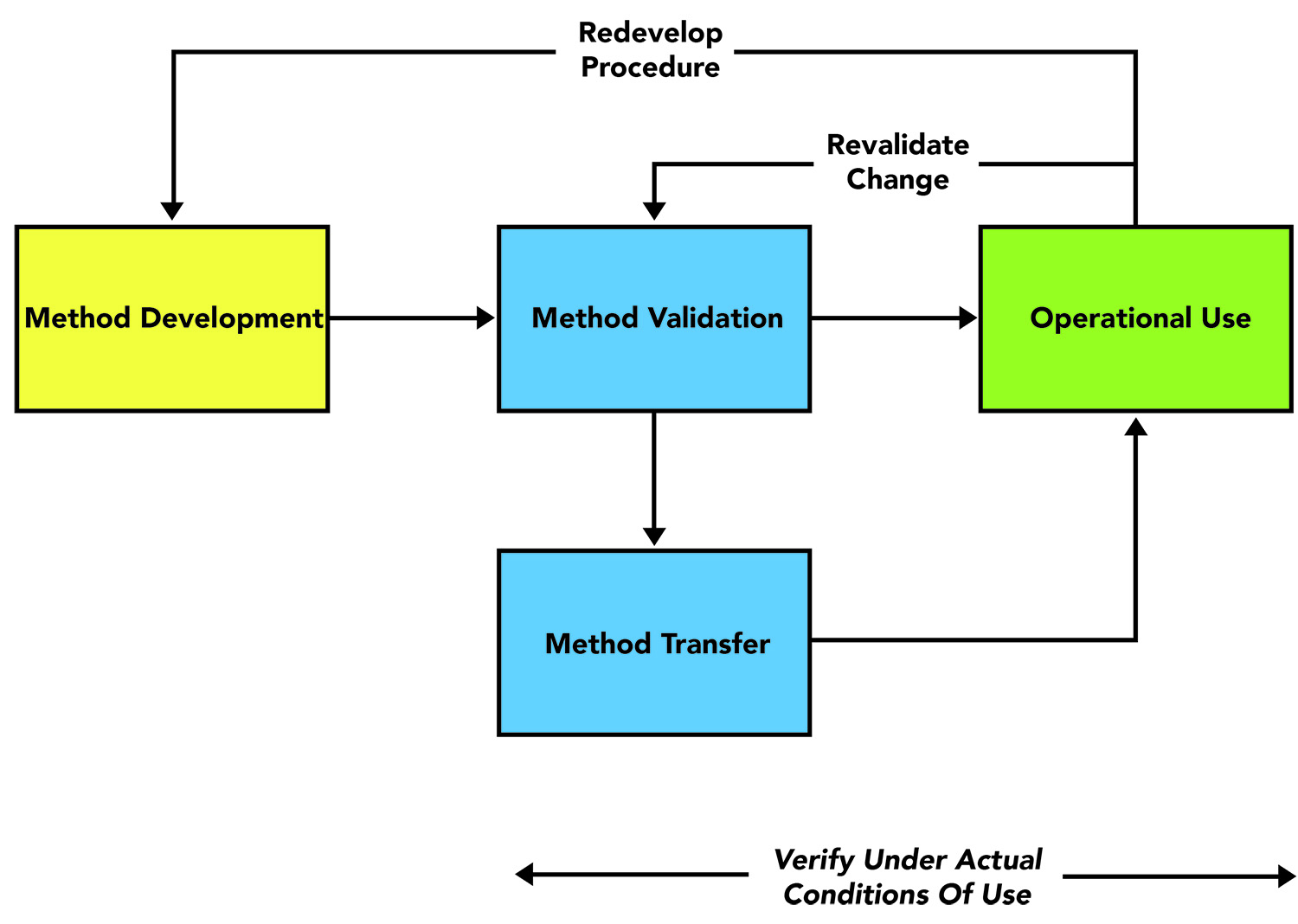
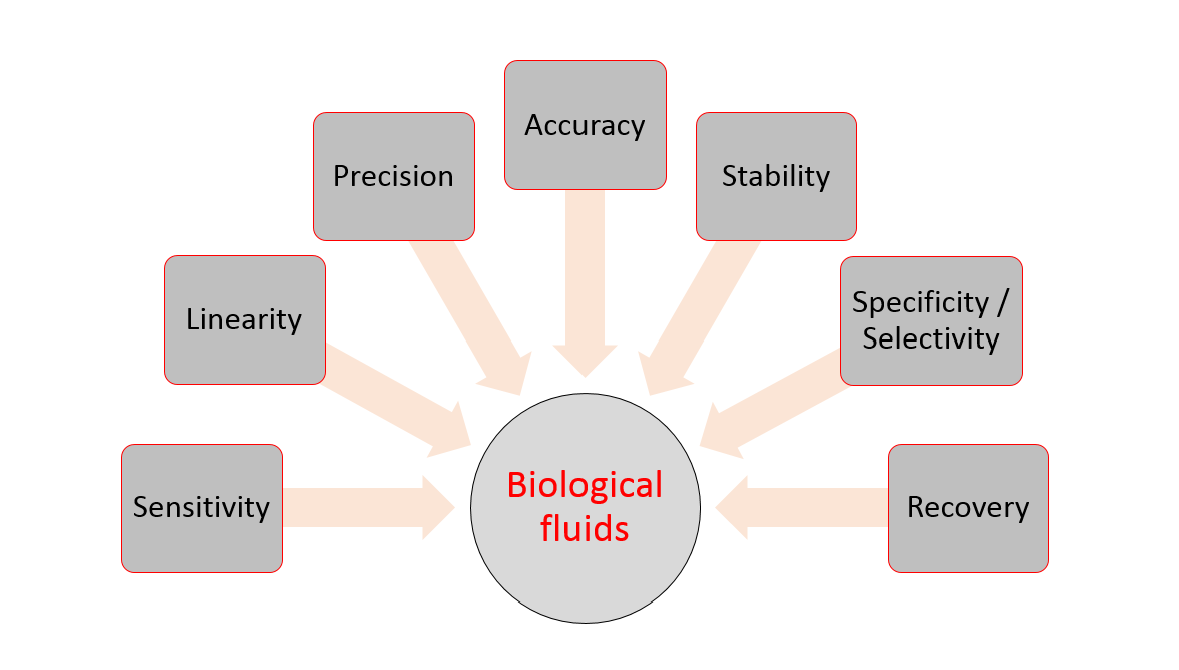
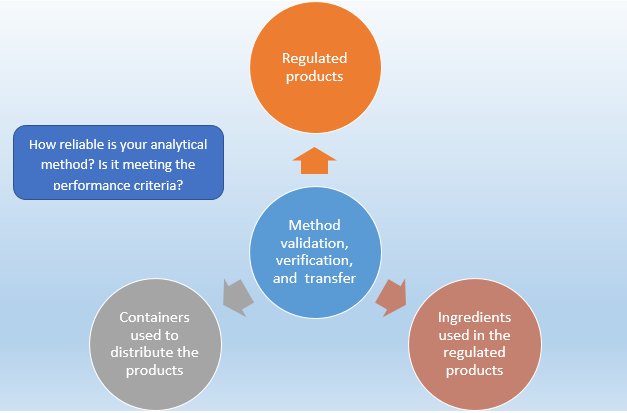
The EMA Bioanalytical Method Validation Guideline: process, history, discussions and evaluation of its content.

PDF) The EMA Bioanalytical Method Validation Guideline: process, history, discussions and evaluation of its content | Peter van Amsterdam - Academia.edu

Bioanalytical method validation: new FDA guidance vs. EMA guideline. Better or worse? | Semantic Scholar

Comparative assessment of bioanalytical method validation guidelines for pharmaceutical industry - ScienceDirect

Validation, Verification and Transfer of Analytical Methods (Understanding and implementing guidelines from FDA/EMA, USP and ICH) (Recorded)
The EMA Bioanalytical Method Validation Guideline: process, history, discussions and evaluation of its content.
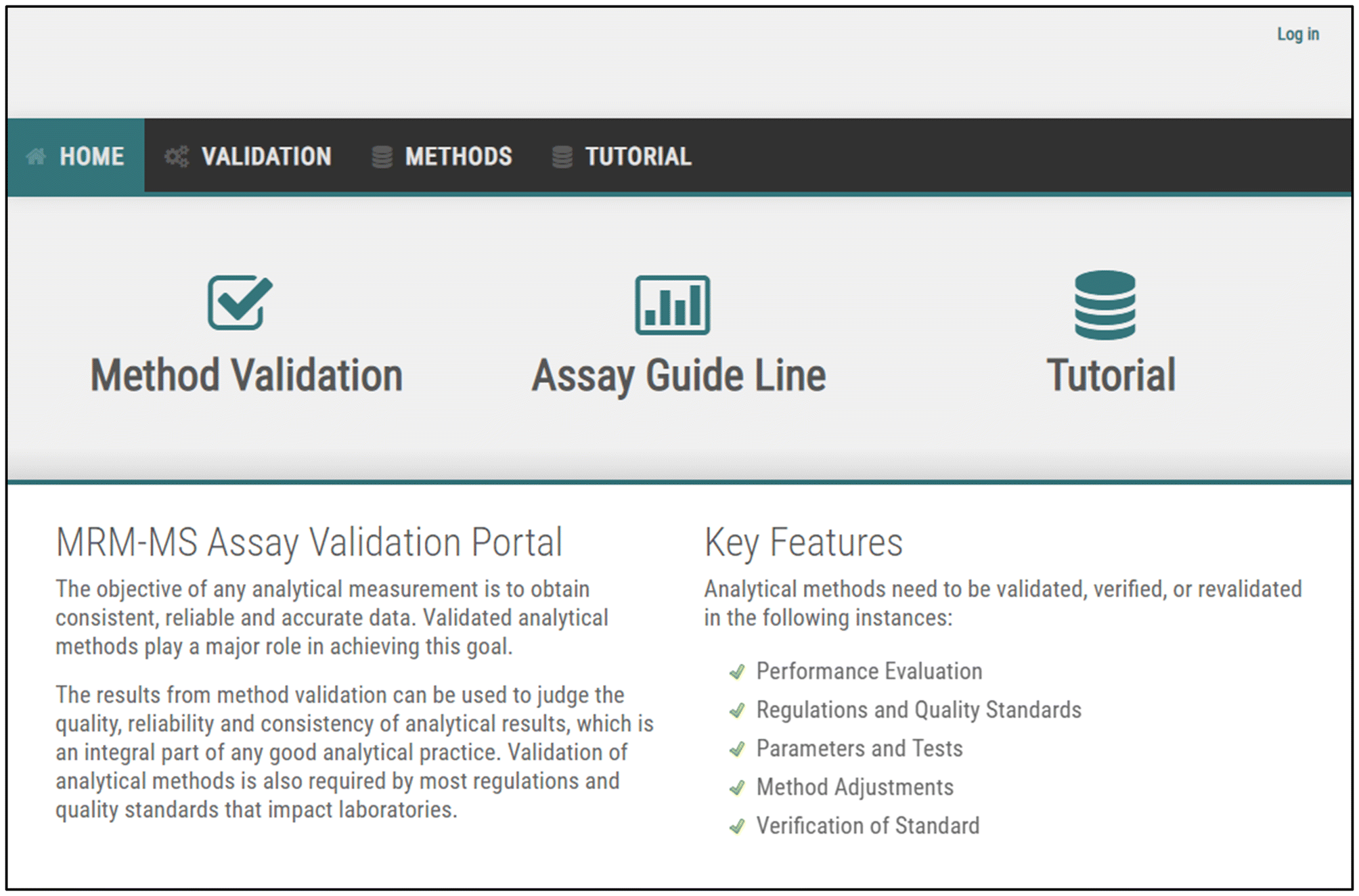
Web portal for analytical validation of MRM-MS assay abided with integrative multinational guidelines | Scientific Reports
![White Paper] Recommendations on the interpretation of the new European Medicines Agency Guideline on Bioanalytical Method Validation by Global CRO Council for Bioanalysis (GCC) - CMIC | Pharmaceutical Development Services (CRO, CDMO, White Paper] Recommendations on the interpretation of the new European Medicines Agency Guideline on Bioanalytical Method Validation by Global CRO Council for Bioanalysis (GCC) - CMIC | Pharmaceutical Development Services (CRO, CDMO,](https://en.cmicgroup.com/wp-content/uploads/2019/11/featuredimage_recommendationsontheinterpretationofnewEMAguidelines.png)
White Paper] Recommendations on the interpretation of the new European Medicines Agency Guideline on Bioanalytical Method Validation by Global CRO Council for Bioanalysis (GCC) - CMIC | Pharmaceutical Development Services (CRO, CDMO,

Frontiers | A Regulatory Risk-Based Approach to ATMP/CGT Development: Integrating Scientific Challenges With Current Regulatory Expectations